Checking for parasite DNA is the best method to monitor the spread of malaria
Researchers reported more people infected with malaria in Equatorial Guinea and Tanzania when they tested using a method that checks for parasite DNA, as compared to what was reported using the common malaria rapid diagnostic tests kits. With the new method, scientists could even detect different types of malaria parasites within a population.
Malaria continues to kill many people in the world. Most doctors use what is called ‘rapid diagnostic tests (RDTs)’ to check for malaria in people. RDTs tests for malaria ‘antigens’ in the blood. Antigens are special proteins that the body produces when it fights a disease.
However, RTDs may not always pick up malaria, especially if patients don’t show any symptoms. Scientists say it is better to check for the parasite DNA instead. DNA are small molecules that contain genetic information.
In this study, the researchers developed and tested a method that checked for malaria parasite DNA when testing for malaria.
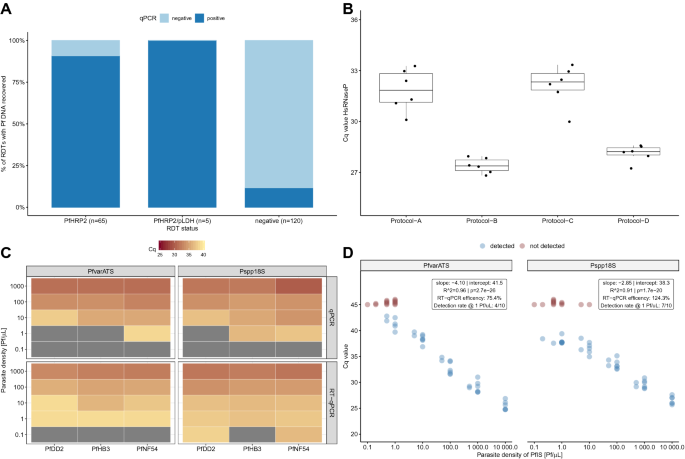
They used blood samples collected from used RDTs kits that had been used in another study that tested for malaria in Bioko Island, Equatorial Guinea in 2018 and Bagamoyo, Tanzania, in 2016 and 2018. They also included Mkuranga district primary school children in Tanzania.
From these blood sample remains, the researchers extracted and tested for malaria parasite DNA.
They said that among Tanzanian primary school children who did not show any symptoms of malaria, their method detected malaria parasite DNA in 91% of positive malaria RDTs, and 12% negative malaria RDTs.
In Bagamoyo, Tanzania, their method detected malaria parasites in 54% of the total samples, while with RDTs only 12% of the samples were said to be positive for malaria.
In Equatorial Guinea, the researchers reported malaria parasite DNA in 31% of people (samples) who did not show symptoms of malaria. They said they were also able to identify 3 different types of malaria parasites. The researchers said unlike what was found in 1996 and 1998 surveys, they did not find the malaria parasite ‘P. vivax’ in Bioko Island. They instead only found ‘P. malariae’ and ‘P. ovale’.
They also said that 1 out of 290 malaria parasites tested was more likely to be resistant to malaria medicine called ‘artemisinin’, meaning that the parasite survives a drug meant to kill it.
The researchers said their method worked well because they did not need lots of blood, and that even after storing samples for over 18 months at room temperature (25 oC), they could still detect malaria parasite DNA.
This study was done in Tanzania and Equatorial Guinea, by researchers based in Switzerland, Equatorial Guinea and Tanzania.
Abstract
The use of malaria rapid diagnostic tests (RDTs) as a source for nucleic acids that can be analyzed via nucleic acid amplification techniques has several advantages, including minimal amounts of blood, sample collection, simplified storage and shipping conditions at room temperature. We have systematically developed and extensively evaluated a procedure to extract total nucleic acids from used malaria RDTs. The co-extraction of DNA and RNA molecules from small volumes of dried blood retained on the RDTs allows detection and quantification of P. falciparum parasites from asymptomatic patients with parasite densities as low as 1 Pf/µL blood using reverse transcription quantitative PCR. Based on the extraction protocol we have developed the ENAR (Extraction of Nucleic Acids from RDTs) approach; a complete workflow for large-scale molecular malaria surveillance. Using RDTs collected during a malaria indicator survey we demonstrated that ENAR provides a powerful tool to analyze nucleic acids from thousands of RDTs in a standardized and high-throughput manner. We found several, known and new, non-synonymous single nucleotide polymorphisms in the propeller region of the kelch 13 gene among isolates circulating on Bioko Island, Equatorial Guinea.
Disclaimer
This summary is a free resource intended to make African research and research that affects Africa, more accessible to non-expert global audiences. It was compiled by ScienceLink's team of professional African science communicators as part of the Masakhane MT: Decolonise Science project. ScienceLink has taken every precaution possible during the writing, editing, and fact-checking process to ensure that this summary is easy to read and understand, while accurately reporting on the facts presented in the original research paper. Note, however, that this summary has not been fact-checked or approved by the authors of the original research paper, so this summary should be used as a secondary resource. Therefore, before using, citing or republishing this summary, please verify the information presented with the original authors of the research paper, or email [email protected] for more information.